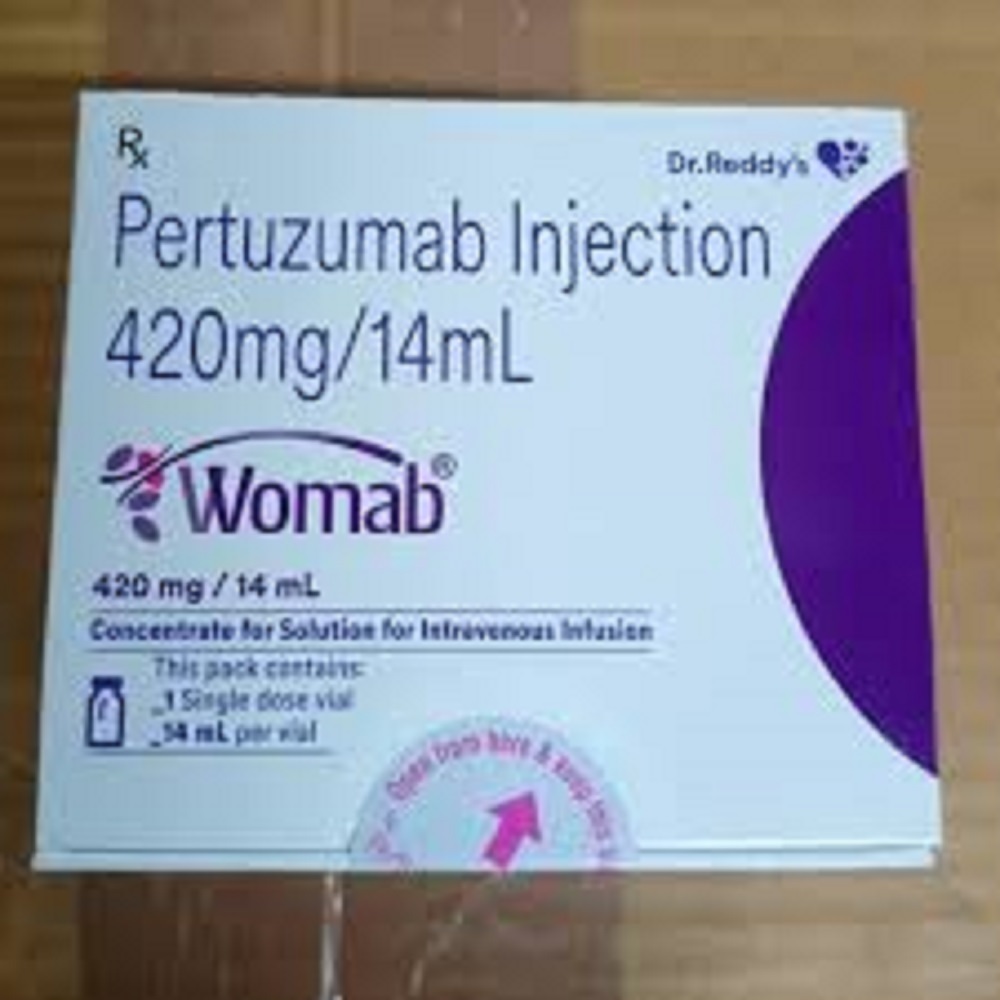
Womab 420 Mg Injection
59000.0 INR/Vial
Product Details:
- Salt Composition Trastuzumab
- Dosage Form Lyophilized powder for solution
- Indication Breast cancer, Gastric cancer (HER2-positive)
- Pacakaging (Quantity Per Box) 1 Vial per box
- Origin of Medicine Imported
- Drug Type General Medicines
- Ingredients Trastuzumab (420mg)
- Click to View more
X
Womab 420 Mg Injection Price And Quantity
- 59000.0 INR/Vial
- 1 Vial
- Prescription
- Intravenous (IV) infusion
- Womab Pharmaceuticals
- Womab
- White to off-white lyophilized powder
- 420 mg/vial
- To be reconstituted before administration; for use in hospital or clinical setting only
Womab 420 Mg Injection Product Specifications
- Trastuzumab
- Lyophilized powder for solution
- General Medicines
- Adults with HER2-positive breast cancer
- Breast cancer, Gastric cancer (HER2-positive)
- Adults
- Administered intravenously under supervision of a healthcare professional
- As directed by physician
- 1 Vial per box
- Liquid
- Imported
- Store in a refrigerator (2 C - 8 C). Do not freeze.
- Trastuzumab (420mg)
- Prescription
- Intravenous (IV) infusion
- Womab Pharmaceuticals
- Womab
- White to off-white lyophilized powder
- 420 mg/vial
- To be reconstituted before administration; for use in hospital or clinical setting only
Womab 420 Mg Injection Trade Information
- 100 Vial Per Month
- 7 Days
Product Description
Hair loss, diarrhea, nausea, and fatigue are some common side effects of this medicine. This medicine may reduce the number of blood cells (decrease white blood cells) in your blood, thereby, increasing the susceptibility to infections. Regular blood tests are required to check your blood cells along with the heart, liver, and kidney.
Many other medicines can affect, or be affected by, this medicine so let your healthcare team know all medications you are using. This medicine is not recommended during pregnancy or while breastfeeding. The use of effective contraception by both males and females during treatment is important to avoid pregnancy.
Targeted Therapy for HER2-Positive Cancer
Womab 420 Mg Injection utilizes Trastuzumab, offering a precise treatment approach for patients diagnosed with HER2-positive breast and gastric cancer. By targeting specific cancer cell receptors, it improves treatment outcomes and helps minimize the risk of damage to healthy cells, making it a significant advancement in oncology therapeutics.
Strict Administration and Handling Requirements
This product is administered as an intravenous infusion, ensuring optimal absorption and therapeutic efficacy. Given its complex preparation, Womab 420 Mg Injection must be reconstituted just before use and handled exclusively by qualified healthcare professionals in controlled clinical environments, promoting patient safety.
Preservation and Packaging for Efficacy
Packaged as a white to off-white lyophilized powder in a single 420 mg vial, Womab requires careful refrigerated storage (2C to 8C) and should never be frozen. Each box contains one vial, guaranteeing consistent dosing and maintaining the integrity of the medicine from manufacturer to patient.
FAQ's of Womab 420 Mg Injection:
Q: How is Womab 420 Mg Injection administered to patients?
A: Womab 420 Mg Injection is given as an intravenous (IV) infusion in hospital or clinical settings. A trained healthcare professional prepares the solution by reconstituting the lyophilized powder and then administers it directly into the patient's bloodstream as directed by a physician.Q: What is the primary benefit of using Womab 420 Mg Injection?
A: This medication provides targeted therapy for HER2-positive breast and gastric cancer. By acting specifically on cancer cells with HER2 overexpression, it offers improved protection for healthy tissues and significantly enhances treatment effectiveness.Q: When should Womab 420 Mg Injection be used?
A: The injection should be used as prescribed by a physician, typically for adult female patients diagnosed with HER2-positive breast or gastric cancer. Administration is supervised and scheduled according to the patient's individual treatment plan and response to therapy.Q: Where should Womab 420 Mg Injection be stored before use?
A: Womab 420 Mg Injection should be kept in a refrigerator at temperatures between 2C and 8C, ensuring the medication's stability. It must never be frozen and should remain in its original packaging until prepared for use.Q: What is the process for preparing Womab 420 Mg Injection before administration?
A: Before administration, the white to off-white lyophilized powder is reconstituted with a specific amount of sterile water for injection by a healthcare professional. The resulting solution is then checked for clarity and used for intravenous infusion.Q: Who is recommended to receive Womab 420 Mg Injection?
A: This injection is suitable for adult female patients diagnosed with HER2-positive breast or gastric cancer. The dosage and frequency are determined by a physician based on individual patient needs and treatment goals.Q: Is Womab 420 Mg Injection a prescription-only medication?
A: Yes, Womab 420 Mg Injection is available only with a prescription and must be administered under the supervision of a qualified healthcare professional in a hospital or clinical environment for safety and efficacy.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email