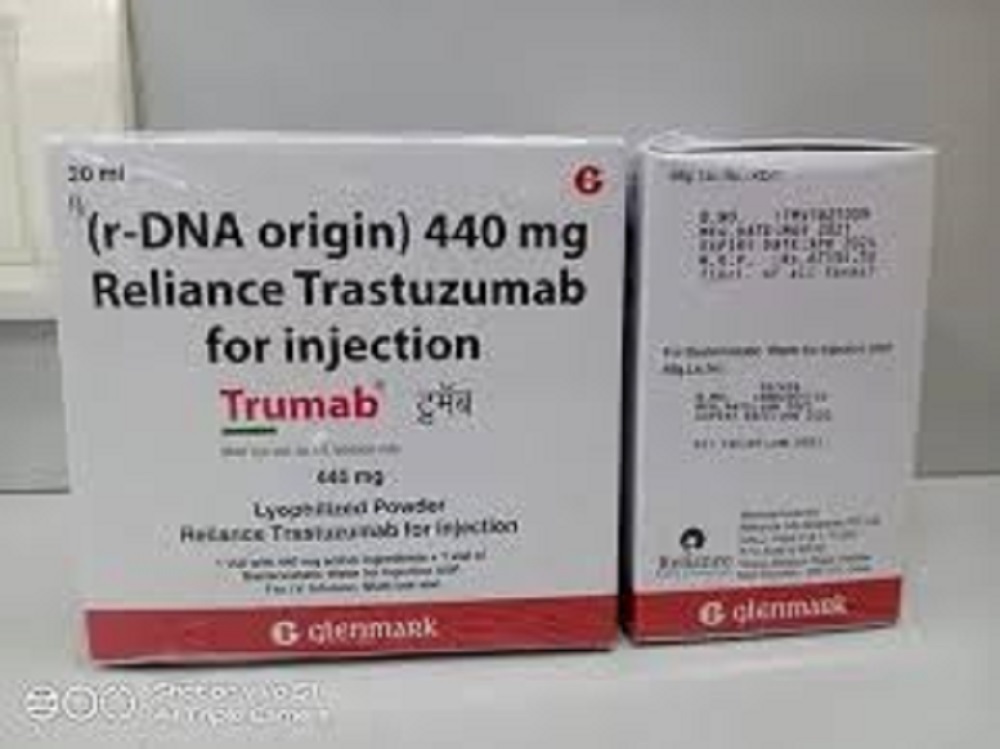
Trumab Trastuzumab 440 Mg Injection
Product Details:
- Dosage Form Injection
- Pacakaging (Quantity Per Box) 1 Vial + Sterile Water for Injection (Bacteriostatic Water for Injection USP 20 mL)
- Indication Treatment of HER2-positive breast and gastric cancers
- Salt Composition Trastuzumab 440 mg
- Origin of Medicine Biological
- Drug Type Prescription
- Ingredients Trastuzumab
- Click to View more
Trumab Trastuzumab 440 Mg Injection Price And Quantity
- 1 Vial
- 9000 INR/Liter
- Yes
- For hospital use only; to be used under medical supervision.
- Intravenous infusion
- Hetero Healthcare Limited
- 24 months from date of manufacture
- Known hypersensitivity to trastuzumab or any component of the formulation
- 440 mg/vial
- Requires reconstitution with provided solvent
- Trumab
Trumab Trastuzumab 440 Mg Injection Product Specifications
- 1 Vial per Box
- Trastuzumab
- Prescription
- Treatment of HER2-positive breast and gastric cancers
- Lyophilized Powder for Injection
- HER2-positive metastatic breast cancer, HER2-positive metastatic gastric cancer
- Injection
- Store at 2C to 8C. Do not freeze.
- 440 mg per vial
- 1 Vial + Sterile Water for Injection (Bacteriostatic Water for Injection USP 20 mL)
- Biological
- To be administered by a healthcare professional as per prescribed schedule
- Anticancer (Monoclonal Antibody)
- Adults
- Trastuzumab 440 mg
- Yes
- For hospital use only; to be used under medical supervision.
- Intravenous infusion
- Hetero Healthcare Limited
- 24 months from date of manufacture
- Known hypersensitivity to trastuzumab or any component of the formulation
- 440 mg/vial
- Requires reconstitution with provided solvent
- Trumab
Product Description
Trumab Injection is given as an injection by a qualified medical professional. Your doctor will decide what dose is necessary and how often you need to take it. This will depend on what you are being treated for and may change from time to time. You should take it exactly as your doctor has advised. Taking it in the wrong way or taking too much can cause very serious side effects. It may take several weeks or months for you to see or feel the benefits but do not stop taking it unless your doctor tells you to.
The most common side effects of this medicine include nausea, headache, rash, insomnia, and infection. This medicine may reduce the number of blood cells (decrease red blood and white blood cells) in your blood, thereby, increasing the susceptibility to infections. Regular blood tests are required to check your blood cells along with heart, liver, and blood uric acid levels. You must have to inform your doctor if you experience breathlessness, cough, shivering, swelling of legs and arm.
Many other medicines can affect, or be affected by, this medicine so let your healthcare team know all medications you are using. This medicine is not recommended during pregnancy or while breastfeeding. The use of effective contraception by both males and females during treatment is important to avoid pregnancy.
Targeted Therapy for HER2-Positive Cancers
Trumab is specifically designed for adults diagnosed with HER2-positive metastatic breast and gastric cancers. As a targeted biological medicine, it binds to the HER2 receptor, helping restrict the growth and spread of cancer cells. Its efficacy and precision make it a preferred choice for oncologists managing these aggressive cancers.
Administration and Dosage Guidelines
This product requires reconstitution with bacteriostatic water provided in the box. The solution must be administered intravenously by qualified healthcare professionals in a hospital setting. Dosage and infusion frequency are determined exclusively by the prescribing oncologist, based on the patient's specific clinical presentation.
FAQ's of Trumab Trastuzumab 440 Mg Injection:
Q: How should Trumab Trastuzumab 440 Mg Injection be prepared and administered?
A: Trumab must be reconstituted using the sterile water included in the package by a qualified healthcare professional. After preparation, it is administered via intravenous infusion in a clinical setting as per the prescribed schedule.Q: What conditions is Trumab recommended to treat?
A: Trumab is indicated for the treatment of adults with HER2-positive metastatic breast cancer and HER2-positive metastatic gastric cancer, where overexpression of the HER2 receptor has been confirmed.Q: When should Trumab not be used?
A: Trumab is contraindicated in individuals with known hypersensitivity to trastuzumab or any component of the formulation. It should not be administered outside of hospital environments nor without medical supervision.Q: Where should Trumab be stored for optimal stability?
A: Trumab should be stored between 2C and 8C and must not be frozen. This ensures the medicine remains effective during its shelf life, which is 24 months from the date of manufacture.Q: What benefits does Trumab offer in treating HER2-positive cancers?
A: As a targeted therapeutic, Trumab binds to the HER2 protein, helping to inhibit cancer cell growth and proliferation. This approach can result in improved outcomes with a manageable safety profile compared to traditional chemotherapy.Q: What is the process for obtaining and using Trumab Trastuzumab 440 Mg Injection?
A: Trumab can only be bought and administered with a prescription from a licensed physician. It is supplied as a single vial per box, along with sterile water, and must be infused in a hospital under the supervision of trained staff.Q: Who is eligible to use Trumab Trastuzumab 440 Mg Injection?
A: The injection is recommended for adult patients diagnosed with HER2-positive metastatic breast or gastric cancer, as evaluated and prescribed by an oncologist.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+