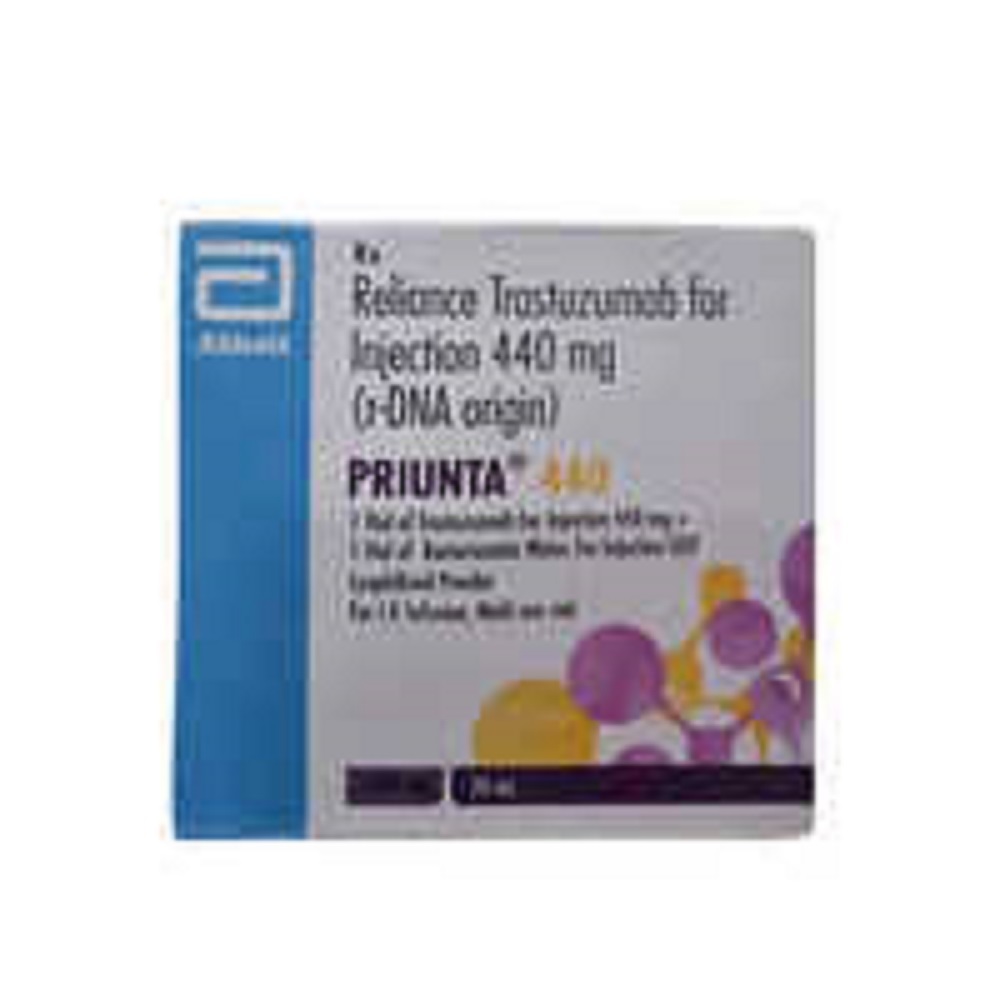
Priunta Trastuzumab 440 Mg Injection
Product Details:
- Indication Metastatic breast cancer, early breast cancer, and metastatic gastric cancer with HER2 overexpression
- Origin of Medicine Biotechnology-derived
- Dosage Form Injection (for intravenous infusion after reconstitution)
- Pacakaging (Quantity Per Box) 1 Vial + 1 ampoule of bacteriostatic water for injection
- Salt Composition Trastuzumab 440mg
- Drug Type Monoclonal Antibody Injection
- Ingredients Trastuzumab
- Click to View more
Priunta Trastuzumab 440 Mg Injection Price And Quantity
- 12000 INR/Piece
- 1 Piece
- Priunta
- White to off-white lyophilized powder
- Bacteriostatic Water for Injection (included)
- Intravenous Infusion
- L01XC03
- 24 months from date of manufacture
- Use only under medical supervision. Monitor cardiac function before and during treatment.
- Anti-neoplastic
- Vial (glass vial with rubber stopper and flip-off seal)
- Prescription Only
Priunta Trastuzumab 440 Mg Injection Product Specifications
- Anti-cancer (HER2 receptor antagonist monoclonal antibody)
- Monoclonal Antibody Injection
- Biotechnology-derived
- Adults
- Store between 2C and 8C. Do not freeze.
- Metastatic breast cancer, early breast cancer, and metastatic gastric cancer with HER2 overexpression
- Trastuzumab 440mg
- Lyophilized Powder for Injection
- Breast and gastric cancer patients overexpressing HER2
- 1 Vial per Box
- 440 mg per vial (reconstituted with 20 ml bacteriostatic water for injection)
- Trastuzumab
- 1 Vial + 1 ampoule of bacteriostatic water for injection
- Injection (for intravenous infusion after reconstitution)
- As directed by the oncologist based on patient body weight and indication
- Priunta
- White to off-white lyophilized powder
- Bacteriostatic Water for Injection (included)
- Intravenous Infusion
- L01XC03
- 24 months from date of manufacture
- Use only under medical supervision. Monitor cardiac function before and during treatment.
- Anti-neoplastic
- Vial (glass vial with rubber stopper and flip-off seal)
- Prescription Only
Product Description
Product introduction
Priunta 440 Injection is used in the treatment of breast and stomach cancer. It works by killing the cancer cells by inhibiting HER2 (human epidermal growth factor receptor protein).
Priunta 440 Injection is given as an injection by a qualified medical professional. Your doctor will decide what dose is necessary and how often you need to take it. This will depend on what you are being treated for and may change from time to time. You should take it exactly as your doctor has advised. Taking it in the wrong way or taking too much can cause very serious side effects. It may take several weeks or months for you to see or feel the benefits but do not stop taking it unless your doctor tells you to.
The most common side effects of this medicine include nausea, headache, rash, insomnia, and infection. This medicine may reduce the number of blood cells (decrease red blood and white blood cells) in your blood, thereby, increasing the susceptibility to infections. Regular blood tests are required to check your blood cells along with heart, liver, and blood uric acid levels. You must have to inform your doctor if you experience breathlessness, cough, shivering, swelling of legs and arm.
Targeted HER2 Therapy for Cancer Care
Priunta Trastuzumab is specifically formulated for adult patients with metastatic and early breast cancer, as well as metastatic gastric cancer, characterized by overexpression of the HER2 protein. Its targeted mechanism offers a more personalized therapy option, aiming to impede cancer cell proliferation while minimizing harm to healthy cells.
Preparation and Safe Administration
Each vial contains 440 mg of trastuzumab lyophilized powder, reconstituted with 20 ml of bacteriostatic water for injection included in the packaging. Administration is by intravenous infusion only, ensuring the medication is delivered directly into the bloodstream for optimal efficacy. Dosing is individualized based on body weight and cancer indication, strictly under an oncologist's guidance.
Guidelines for Handling and Storage
Priunta should be stored at 2C to 8C and never frozen. The glass vial comes with a rubber stopper and a flip-off seal for product integrity. Use the reconstituted solution promptly as instructed by a healthcare provider to ensure stability and potency. Routine cardiac evaluation is advised before treatment initiation and throughout therapy.
FAQ's of Priunta Trastuzumab 440 Mg Injection:
Q: How is Priunta Trastuzumab 440 mg Injection administered?
A: Priunta Trastuzumab is administered intravenously following reconstitution with the included bacteriostatic water for injection. Qualified healthcare professionals infuse the solution under controlled clinical settings.Q: What cancers is Priunta Trastuzumab indicated to treat?
A: Priunta is indicated for metastatic and early breast cancer, as well as metastatic gastric cancer, in patients whose tumors overexpress the HER2 protein.Q: When should cardiac function be monitored during Priunta therapy?
A: Cardiac function must be evaluated before starting therapy and regularly during treatment, as trastuzumab may affect heart health.Q: Where should Priunta Injection be stored before use?
A: Priunta should be refrigerated between 2C and 8C. It must not be frozen, and should be kept in its original packaging until ready for use.Q: What is the process for preparing Priunta Trastuzumab for infusion?
A: The lyophilized powder is reconstituted with 20 ml bacteriostatic water for injection (provided), by a trained healthcare professional, and then prepared for intravenous infusion as per medical direction.Q: How is the correct dosage of Priunta determined?
A: Dosage is calculated by the prescribing oncologist based on the patient's body weight, overall health status, and specific cancer indication.Q: What are the main benefits of using Priunta Trastuzumab in eligible patients?
A: Priunta offers a targeted treatment approach for HER2-positive cancers, potentially improving outcomes, reducing the likelihood of cancer progression, and providing a personalized therapy option tailored to specific tumor characteristics.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+