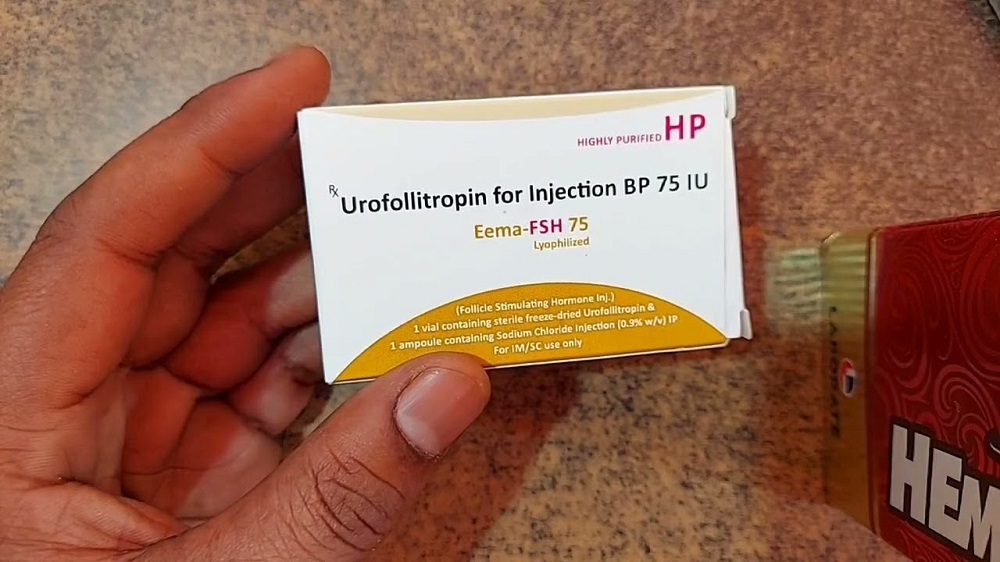
Eema Fsh 75 Iu Injection
Product Details:
- Salt Composition Follicle Stimulating Hormone (FSH) 75 IU
- Pacakaging (Quantity Per Box) 1 single-use vial per box
- Origin of Medicine Allopathic
- Dosage Form Lyophilized Powder for Injection
- Indication Anovulation, Ovarian Dysfunction, Infertility due to insufficient FSH
- Drug Type Hormonal Injection
- Ingredients Highly Purified Recombinant Follicle Stimulating Hormone (FSH)
- Click to View more
Eema Fsh 75 Iu Injection Price And Quantity
- 820 INR/Vial
- 1 Vial
- Requires reconstitution with provided solvent before administration
- Hypersensitivity to FSH, uncontrolled thyroid/adrenal dysfunction, pregnancy
- Monitor ovarian response with ultrasound, avoid in cases of ovarian cyst unrelated to PCOS
- Abdominal pain, headache, ovarian enlargement, injection site reaction
- Gonadotropin Hormone
- Clear to slightly opalescent once reconstituted
- Eema Fsh 75 Iu Injection
- 24 months from manufacturing date
- Subcutaneous Injection
- Prescription Only
Eema Fsh 75 Iu Injection Product Specifications
- Anovulation, Ovarian Dysfunction, Infertility due to insufficient FSH
- 1 Vial
- Lyophilized Powder for Injection
- 75 IU per vial, as directed by physician
- Hormonal Injection
- Allopathic
- Store in refrigerator (2C8C). Do not freeze. Protect from light.
- Highly Purified Recombinant Follicle Stimulating Hormone (FSH)
- 1 single-use vial per box
- Injection
- Adults (Female)
- Female infertility treatment, ovulation induction, assisted reproductive technology (ART)
- Administered subcutaneously; dosage and duration as prescribed by physician
- Follicle Stimulating Hormone (FSH) 75 IU
- Stimulates ovarian follicle growth and development
- Requires reconstitution with provided solvent before administration
- Hypersensitivity to FSH, uncontrolled thyroid/adrenal dysfunction, pregnancy
- Monitor ovarian response with ultrasound, avoid in cases of ovarian cyst unrelated to PCOS
- Abdominal pain, headache, ovarian enlargement, injection site reaction
- Gonadotropin Hormone
- Clear to slightly opalescent once reconstituted
- Eema Fsh 75 Iu Injection
- 24 months from manufacturing date
- Subcutaneous Injection
- Prescription Only
Product Description
EMA FSH 75IU INJECTION belongs to the class of drugs called hormones used in the treatment of female infertility in women whose ovaries do not produce eggs and in women receiving treatments like IVF (in-vitro fertilization). Infertility is unable to become pregnant after trying for a year. Female infertility may be caused due to different factors affecting ovulation (release of an egg from the ovary), fertilization (fusion of sperm and egg) and implantation (fertilized egg attaches to uterus lining).
EEMA FSH 75IU INJECTION contains Urofollitropin which works similarly to follicle-stimulating hormone (natural sex hormone). EEMA FSH 75IU INJECTION increases oestrogen production that stimulates the development of the egg and mature follicle in women having ovulation (release of an egg from the ovary) problems and those undergoing IVF treatments.
Effective Solution for Female Infertility
Eema Fsh 75 IU Injection offers targeted stimulation of ovarian follicles in women diagnosed with anovulation or ovarian dysfunction. By supporting the natural ovulation process, this therapy assists with increasing the likelihood of conception, especially in assisted reproductive technology (ART) cycles. This injection is strictly administered under medical supervision to ensure optimal outcomes and minimize potential risks.
Proper Reconstitution and Administration
This injection is supplied as a lyophilized powder and must be reconstituted with the provided sterile solvent before administration. Once reconstituted, the solution should appear clear to slightly opalescent. It is injected subcutaneously as directed by a healthcare provider. Proper preparation and administration are essential for maximizing therapeutic effectiveness and safety during treatment.
Essential Safety and Monitoring Guidelines
Use of Eema Fsh 75 IU Injection mandates careful monitoring, including regular ultrasound examinations to assess ovarian response. The medication is contraindicated in women with hypersensitivity to FSH, uncontrolled thyroid or adrenal dysfunction, current pregnancy, or certain ovarian cysts. Adverse effects such as headache, abdominal pain, and injection site reactions should be promptly reported to the physician.
FAQ's of Eema Fsh 75 Iu Injection:
Q: How is Eema Fsh 75 IU Injection administered?
A: Eema Fsh 75 IU Injection is administered subcutaneously (under the skin) after reconstituting the lyophilized powder with the provided solvent. The injection should only be given as directed by your physician, typically in a clinical setting or at home after adequate training.Q: What are the primary uses and benefits of Eema Fsh 75 IU Injection?
A: This medication is used to treat female infertility by stimulating the growth and development of ovarian follicles. It benefits women undergoing ovulation induction or assisted reproductive technology (ART) due to anovulation or insufficient FSH production.Q: When should Eema Fsh 75 IU Injection not be used?
A: Avoid using this injection if you have a known allergy to FSH, uncontrolled thyroid or adrenal disorders, are pregnant, or have ovarian cysts unrelated to polycystic ovary syndrome (PCOS). Always inform your doctor of your complete medical history before starting treatment.Q: What is the process for preparing Eema Fsh 75 IU Injection for use?
A: To prepare, gently reconstitute the powder in the vial with the supplied sterile solvent until it appears clear to slightly opalescent. Use the solution immediately after preparation, following your physician's instructions for correct injection technique.Q: What precautions should be taken during therapy with Eema Fsh 75 IU Injection?
A: Close monitoring of ovarian response via ultrasound is essential to minimize risk of ovarian hyperstimulation. Notify your healthcare provider if you experience side effects such as abdominal pain, headaches, or injection site reactions. Do not use if pregnant or with certain ovarian conditions.Q: Where should Eema Fsh 75 IU Injection be stored before use?
A: Store the injection vial in a refrigerator at 2C-8C. Do not freeze the medication and always protect it from direct light to maintain its effectiveness until the expiry date.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+